READ THE FULL TNXP RESEARCH REPORT
Financial Update
On July 25, 2025, Tonix Pharmaceuticals Holding Corp. (NASDAQ:TNXP) filed an 8-K in which it disclosed preliminary operating results for the second quarter of 2025. Net revenue from product sales for the quarter ended June 30, 2025, was approximately $2.0 million, compared to $2.2 million for the quarter ended June 30, 2024. Net operating loss for the second quarter of 2025 was approximately $26.3 million, compared to $78.8 million for the second quarter of 2024. As of July 25, 2025, the company had approximately 8.7 million shares outstanding. Tonix exited the second quarter of 2025 with approximately $125.3 million in cash and cash equivalents. Subsequently, the company raised $50.6 million in cash from equity offerings in July 2025. We estimate the company has sufficient capital to fund operations into the third quarter of 2026. Importantly, the company’s balance sheet also provides plenty of flexibility for the commercial launch of TNX-102 SL, if it is approved by the FDA on or before the PDUFA date of August 15, 2025. If approved, Tonix is planning a sales force for TNX-102 SL of between 70 and 90 sales representatives.
Business Update
Deep Dive into TNX-1500
Tonix is developing TNX-1500, a third-generation anti-CD40 ligand (CD40L, also known as CD154) antibody, for the prevention of allograft and bone marrow transplant rejection and the treatment of autoimmune diseases. The company recently announced positive Phase 1 results for TNX-1500, in which the drug was generally well-tolerated, had a favorable safety profile, and blocked primary and secondary antibody responses to a test antigen. Based on these results and subject to IND approval, Tonix plans to pursue a Phase 2 clinical trial of TNX-1500 for the prevention of kidney transplant rejection. In this report, we provide an overview of TNX-1500 and its potential to prevent organ transplant rejection.
Solid Organ Transplantation
In 2023, more than 46,000 solid organ transplants occurred in the U.S. (OPTN). This represents an 8.7% increase over 2022, with more than 28,000 of those involving renal transplantation. The growing number of transplants is due in part to the use of calcineurin inhibitors, particularly tacrolimus, that are part of the standard of care for reducing cellular and antibody mediated rejection and improving one-year survival rates (Camilleri et al., 2016). Unfortunately, the use of calcineurin inhibitors is associated with an increased risk for both acute and chronic nephrotoxicity in all types of solid transplants (Naesens et al., 2009). Thus, while calcineurin inhibitors have greatly increased the one-year survival rates for organ transplant, the long-term survival rates (>3 years) have not significantly changed.
One strategy to improve the long-term outcomes for solid organ transplant recipients is to inhibit various co-stimulatory pathways of the immune system. The inhibition of numerous co-stimulatory pathways has been attempted, including CD80 and CD86 (Birsan et al., 2003), CD28 (Levisetti et al., 1997), and CD40 and CD40L (Xu et al., 2002). Nulogix (belatacept) is a CTLA-4 Ig fusion protein that blocks both CD80 and CD86 and is approved for prevention of kidney transplant rejection. Based on the generated data, it became apparent that antagonizing the CD40/CD40L pathway has the potential to be more efficacious than inhibition of other pathways.
The CD40/CD40L signaling pathway is involved in the activation of both the innate and adaptive immune response. CD40 is predominantly expressed on antigen presenting cells (APCs) and delivers intracellular activating signals. CD40L, which does not contain any signaling capacity, is found mostly on activated T cells, but is also found on other cell types, including B cells, natural killer (NK) cells, macrophages, and platelets (Schönbeck et al., 2001). The CD40/CD40L pathway is essential for humoral immune responses to T cell-dependent antigens (Lederman et al., 1992), the production of proinflammatory cytokines (Cella et al., 1996), and generating effective cytotoxic T cell responses (Liu et al., 2013). CD40L is a ligand for both CD40 and CD11b and blocking both interactions is the best means to prevent allotransplant rejection (Liu et al., 2020). Thus, an anti-CD40L antibody may be more efficacious than those targeting CD40.
In support of this, a meta-analysis analyzed nonhuman primate studies that compared anti-CD40 and anti-CD40L antibody treatments for the prevention of renal transplant rejection (Perrin et al., 2022). A total of eleven studies were identified that examined either anti-CD40 or anti-CD40L antibody monotherapy following renal transplantation. The following figure shows the survival across groups between untreated, anti-CD40 and anti-CD40L animals. While both treatments increased the probability of rejection-free survival compared to placebo, anti-CD40L treatment resulted in a median survival of 352 days compared to 131 days for anti-CD40 treatment (P=0.0001).
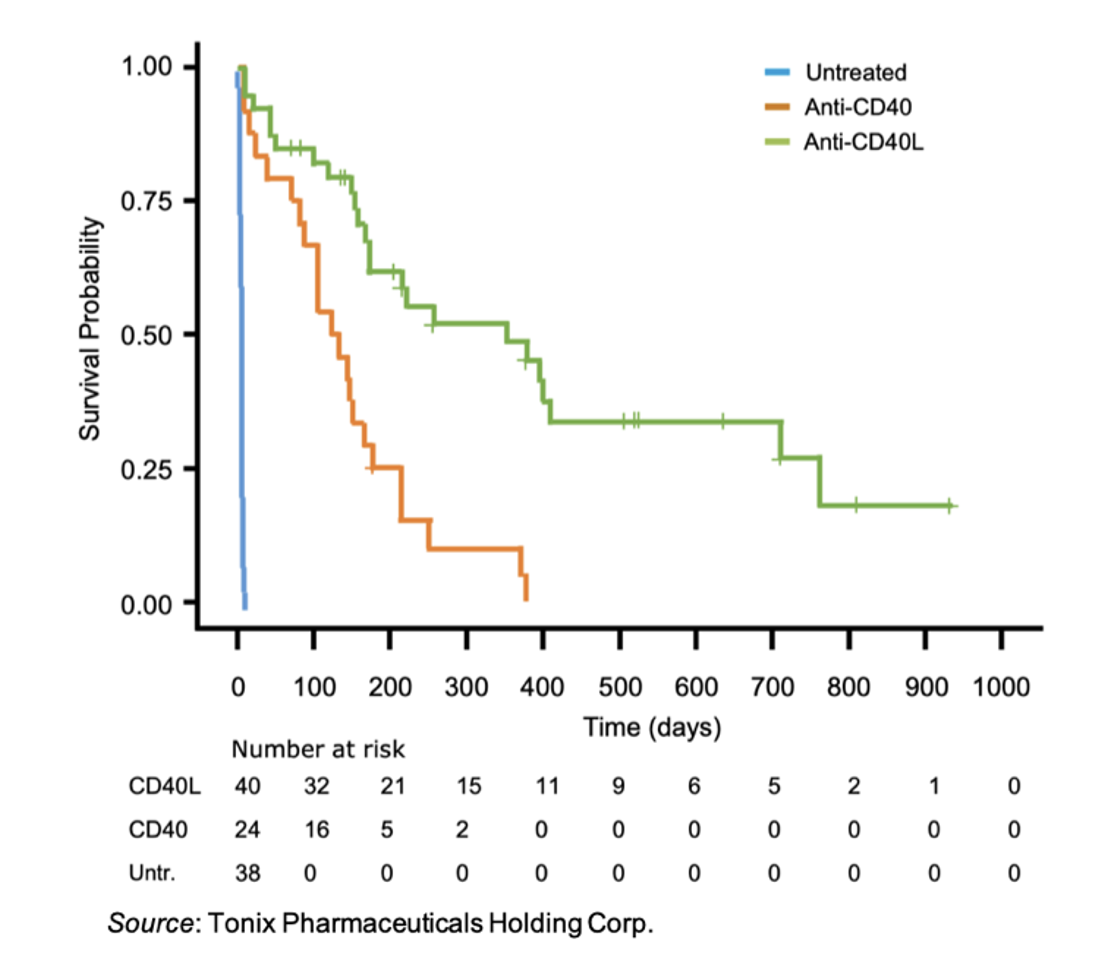
Background on TNX-1500
TNX-1500 is a third-generation anti-CD40L antibody that has been modified to alleviate issues involved in the development of first-generation anti-CD40L antibodies. Ruplizumab (BG9588) was a first-generation anti-CD40L antibody that was not advanced in development after thromboembolic events were noted in an early-stage clinical trial in systemic lupus erythematosus (SLE) patients (Boumpas et al., 2003). The pro-thrombotic effects of anti-CD40L therapy were also seen in non-human primate studies (Kawai et al., 2000). Thus, researchers sought to elucidate the mechanism behind why anti-CD40L therapy led to an increased risk of thromboembolism.
Multiple in vitro studies identified FcγRIIa as the driving force behind the anti-CD40L antibody-mediated platelet activation seen in clinical trials. FcγRIIa (CD32A) is the low-affinity receptor for the constant fragment (Fc) of immunoglobulin (Ig) G (Qiao et al., 2015). Immune complexes, such as those formed between anti-CD40L antibodies and soluble CD40L, can bind FcγRIIa and trigger intracellular signaling events that lead to platelet activation and aggregation. Interestingly, mouse studies were not able to recapitulate the effects of the anti-CD40L studies, most likely due to the fact that murine platelets do not express FcγRIIa. Seeming to confirm this, a study involving a humanized mouse model in which the human FcγRIIa was expressed on murine platelets showed platelet activation and thrombus formation following administration of preformed immune complexes of soluble CD40L and anti-CD40L antibodies (Robles-Carrillo et al., 2010). Utilizing this information, multiple groups attempted to design anti-CD40L antibodies that lacked FcγRIIa binding, either through elimination of the Fc region of the antibody or mutation of the FcγRIIa binding domain. These are shown in the figure below.
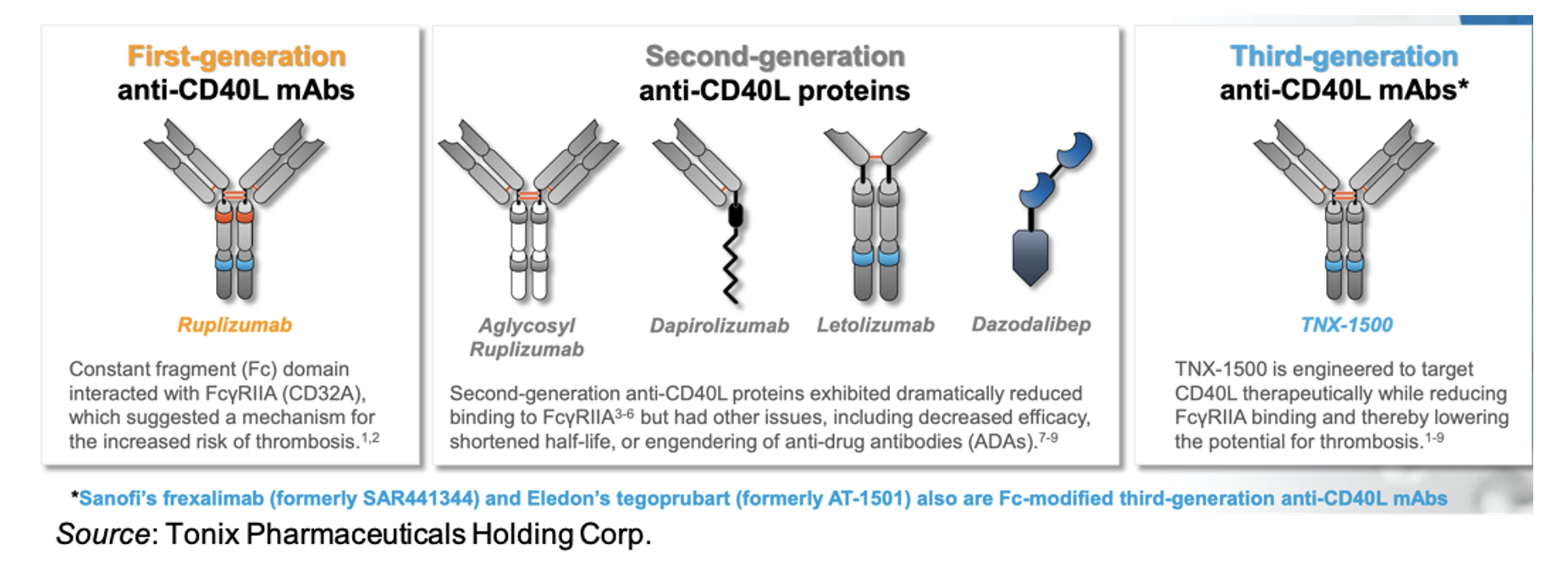
While the “second generation” anti-CD40L antibodies exhibit decreased FcγRIIa binding, additional issues with these molecules exist. For example, an aglycosyl form of ruplizumab that had reduced binding to FcγRIIa was ineffective in non-human primate renal and islet allotransplantation (Ferrant et al., 2004). Another study involving letolizumab showed no thromboembolic events, but required additional immunosuppressive agents to achieve long-term (>200 days) renal allograft survival (Kim et al., 2017).
The following image shows the overall structure of TNX-1500, which consists of the full Fab portion of ruplizumab and a FcγR-modulated Fc region of IgG4 that diminishes binding to FcγRIIa. The antibody includes the full complement of disulfide bonds that bind the two heavy chains together and that link the heavy and light chains. This is in contrast to the other anti-CD40L proteins that either lack the Fc region of the antibody or the full complement of disulfide bonds. Keeping the native antibody structure leads to an increased half-life compared to other anti-CD40L proteins, with pharmacokinetic data supporting once a month dosing.
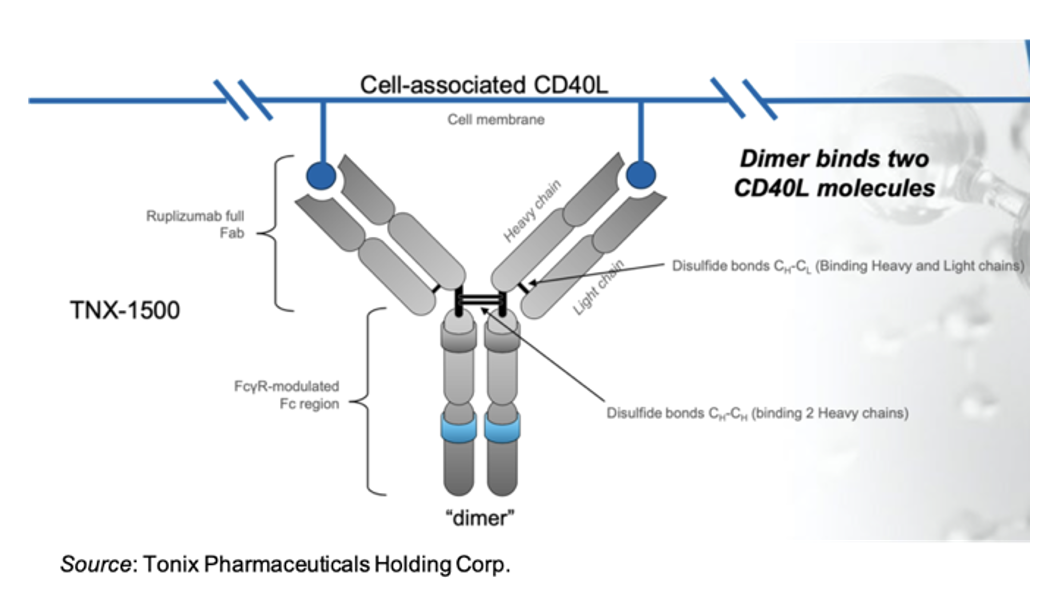
Preclinical Results with TNX-1500
TNX-1500 has been evaluated in multiple preclinical, non-human primate transplant studies, as discussed in the following two papers published in the August 2023 edition of the American Journal of Transplantation:
Lassiter et al., 2023: This study evaluated TNX-1500 both in vitro and in vivo as part of an immunosuppressive regime following renal allograft. TNX1500 in association with soluble CD40L did not activate primate or human platelets, which was in direct contrast to ruplizumab, which did activate platelets in vitro. There were no vascular thrombotic complications in kidney transplant recipients treated with TNX-1500 in the absence of prophylaxis for thrombosis. The absence of thrombosis was not associated with an absence of immunosuppressive efficacy, as 83% of allograft recipients survived to six months. While not statistically significant, there was a strong trend toward improved transplant survival relative to historical animals that were administered the conventional triple drug immunosuppressive cocktail of tacrilomus, mycophenolate mofetil (MMF), and steroids. Lastly, there were no side effects or infectious complications, which are often seen with the conventional immunosuppression therapy.
Miura et al., 2023: This study evaluated TNX-1500 as an immunosuppressive agent following cardiac transplant in non-human primates. The results showed that treatment with TNX-1500 was well tolerated and prevented pathogenic alloimmunity or development of anti-donor antibodies. This led to prolonged survival with preserved graft function. In addition, there appeared to be an increase in regulatory T cells (Tregs) that promoted peripheral regulatory tolerance.
TNX-1500 was also utilized as part of an immunosuppressive regime to prevent rejection in kidney xenograft transplants (Anand et al., 2023). A porcine donor was engineered to carry 69 genomic edits, eliminating glycan antigens, overexpressing human transgenes, and inactivating porcine endogenous retroviruses. The xenograft resulted in long-term (>2 years) survival of the recipient.
Tonix is also collaborating with Boston Children’s Hospital to evaluate TNX-1500 for the prevention of graft-versus-host disease (GvHD) following hematopoietic stem cell transplantation (HCT) in animals. The primary objective of the study is to determine if TNX-1500 administered prophylactically can modify GvHD progression in animals following HCT. If positive results are obtained from this study, they would be used to support an IND filing for clinical trials in bone marrow transplantation.
Clinical Results with TNX-1500
In August 2023, Tonix initiated a Phase 1 single ascending dose trial of TNX-1500 in healthy volunteers. It consisted of three dosing cohorts (3 mg/kg, 10 mg/kg, 30 mg/kg) with six, 10, and 10 subjects (2 placebo per cohort) in each cohort, respectively. TNX-1500 was administered through IV infusion over a period of one hour. Participants were observed in the clinic for one day and then had periodic clinic visits to Day 120. To test the pharmacodynamic properties of TNX-1500, participants received an antigen challenge with keyhole limpet hemocyanin (KLH; Immucothel®) administered subcutaneously on Day 2 and Day 29. Samples to evaluate anti-KLH antibodies were obtained on Day 1, 8, 15, 29, 36, 50, 64, 78, 120. Twenty-six subjects were enrolled into the study and 24 of them completed the study (one placebo participant was lost to follow-up and one on TNX-1500 withdrew consent).
The topline results were announced in February 2025 and showed that TNX-1500 was generally well-tolerated and exhibited a favorable safety and tolerability profile. The only treatment emergent adverse event (TEAEs) that occurred in ≥ 3 participants in the TNX-1500 group was aphthous ulcer that occurred in one participant each in the 3 mg/kg, 10 mg/kg, and 30 mg/kg groups. All were rated as mild, possible related, and resolved in 2-10 days. Importantly, there were no thromboembolic events, which were prespecified as TEAEs of special interest.
The KLH results showed that TNX-1500 at both 10 mg/kg and 30 mg/kg blocked the primary and secondary anti-KLH antibody responses as shown by the mean antibody level at all sampled timepoints being below the lower limit of quantitation (400 mg/L). TNX-1500 at 3 mg/kg blocked the primary response and reduced the peak secondary response to KLH Day 29 challenge by approximately 2/3rd (69%) relative to the peak response to placebo. The mean half-life of TNX-1500 was: 3 mg/kg, 19.6 (9.29) days; 10 mg/kg, 37.8 (5.46) days; 30 mg/kg, 33.7 (4.83) days.
The positive results from the Phase 1 study showed that TNX-1500 may be eligible for once a month dosing based on the half-life data. In addition, as shown by blocking the primary and secondary antibody response to KLH, the Fc modifications that were introduced to alleviate potential thrombotic complications did not attenuate the potency of TNX-1500. These results support advancing TNX-1500 to a Phase 2 trial for the prevention of kidney transplant rejection.
Other CD40L Antibodies in Development
There are multiple anti-CD40L antibodies currently in development, including two other third-generation antibodies.
Frexalimab (INX021) – Sanofi: This is an Fc-modified anti-CD40L antibody that has been studied as a potential therapy for Sjogren’s Syndrome (SjS), multiple sclerosis (MS), and SLE. The program in SjS has been discontinued. Phase 2 results in MS showed that frexalimab treatment resulted in a greater reduction in the number of new gadolinium-enhancing T1-weighted lesions at Week 12 as compared to placebo (Vermersch et al., 2024), however larger and longer trials will be necessary to determine its full efficacy in MS. A Phase 2 trial in SLE has initiated (NCT05039840).
Tegoprubart – Eledon: This is a monoclonal antibody in which four point-substitutions were introduced into the Hu5C8 heavy change hinge and hinge-proximal CH2 constant domain (C220S/C226S/C229S/P238S), with in vitro studies showing no binding of tegoprubart to FcγRI, FcγRIIa, FcγRIIIa, or FcγRIIIb. Tegoprubart lacks disulfide bonds between the heavy and light chains and between the heavy chains (WHO Drug Information, 2022). A Phase 2a trial in ALS was successfully completed and showed the drug was safe and well tolerated and met target engagement in a dose-dependent fashion (Perrin et al., 2024). Eledon is expected to announce topline results from the Phase 2 BESTOW trial in kidney transplantation in the fourth quarter of 2025.
Dapirolizumab – Biogen/UCB: This is an Fc-free polyethylene glycol (PEG)-conjugated antigen-binding (Fab’) fragment that binds to CD40L. In September 2024, Biogen and UCB announced positive topline results from the Phase 3 PHOENYCS GO study in patients with moderate-to-severe systemic lupus erythematosus (SLE). The Phase 3 PHOENYCS FLY study is currently enrolling (NCT06617325).
Dazodalibep – Amgen: This is a non-antibody CD40L antagonist that lacks an Fc region and consists of CD40L binding sites grafted onto a Tn3 scaffold. In vitro studies show dazodalibep does not induce platelet aggregation. Prior to being acquired by Amgen, Horizon Therapeutics reported positive results in two different SjS populations, those with moderate to severe systemic disease activity and those with moderate to severe localized symptoms.
Lu AG22515 – Lundbeck and AprilBio Co., Ltd.: This is a recombinant protein utilizing AprilBio’s SAFA technology that consists of an anti-human serum albumin (HAS) Fab with an anti-CD40L Fab covalently attached. The compound has completed a Phase 1 study in healthy adults and a trial to evaluate its potential in Thyroid Eye Disease has initiated.
Market Opportunity
There are approximately 28,000 kidney transplants performed each year in the U.S. We estimate that the yearly cost for TNX-1500 would be $50,000 per year. Assuming peak market share of 20% leads to estimated peak sales of approximately $275 million. Globally, there were approximately 111,000 kidney transplants in 2023. Assuming 60% of those patients could be targeted, with a 15% market penetration and a yearly cost of $30,000 leads to potential peak sales of approximately $300 million.
In addition to preventing organ transplant rejection, Tonix is also interested in pursuing TNX-1500 as a treatment for autoimmune disease. Potential indications include systemic lupus erythematosus, multiple sclerosis, Sjogren’s Syndrome, and psoriatic arthritis. Each of those indications will require large studies, but offer large market potential. We believe Tonix would likely enter into one or more partnerships before initiating an autoimmune disease program. The global autoimmune disease therapeutics market is currently valued at approximately $124 billion and is estimated to grow to $250 billion by 2034 (Zion Market Research).
Conclusion
While Tonix is fully focused on the upcoming PDUFA date for TNX-102 SL for the management of fibromyalgia on August 15, 2025, and the potential commercial launch of the drug contingent on FDA approval, the company is also continuing the advancement of TNX-1500 as a treatment to prevent organ transplant rejection and potentially as a treatment for certain autoimmune diseases. There is a substantial amount of preclinical data supporting the use of TNX-1500 in the prevention of allograft rejection, and we look forward to updates from the company as it advances TNX-1500 toward a Phase 2 trial in kidney transplant recipients. As we await the PDUFA decision, our valuation for Tonix is currently $50 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.






