READ THE FULL SNGX RESEARCH REPORT
Business Update
Multiple Catalysts Ahead in 2H25
Soligenix, Inc. (NASDAQ:SNGX) is a late-stage biopharmaceutical company developing products to treat rare diseases within two business segments: Specialized BioTherapeutics and Public Health Solutions. The Specialized BioTherapeutics business segment currently has multiple products under development with multiple catalysts anticipated in the second half of 2025.
Phase 3 FLASH2 Trial: Enrollment Updated Anticipated in 2H25
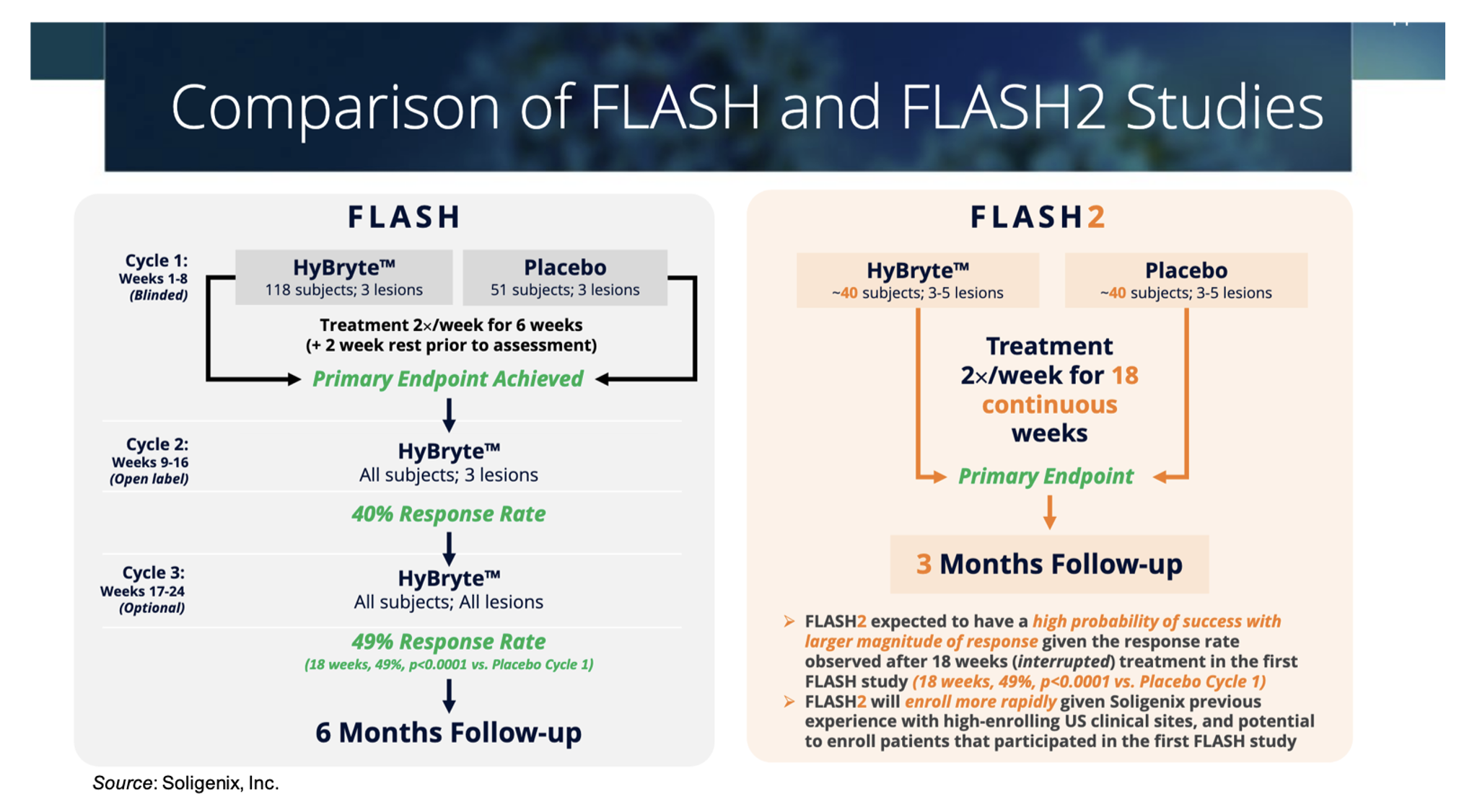
The Phase 3 FLASH2 Trial is evaluating HyBryte™ (SGX301 or synthetic hypericin) in 80 patients with early-stage cutaneous T cell lymphoma (CTCL). The FLASH2 trial is very similar in design to the successful Phase 3 FLASH trial, as shown in the following figure, which provides a comparison between the two studies. One key difference between the trials is that in the FLASH trial patients were treated for three cycles of six-weeks each, with a two-week break in between cycles and the primary efficacy endpoint was measured after the first treatment cycle, while in the FLASH2 trial patients will be treated for 18 consecutive weeks before the primary efficacy endpoint is assessed. Based on the results from the FLASH trial, we believe the FLASH2 trial has a high probability of success.
Investigator-Initiated Study (IIS): Clinical Update in 3Q25
An IIS of HyBryte in CTCL patients is currently being conducted by Ellen Kim, MD, Director, Penn Cutaneous Lymphoma Program, Vice Chair of Clinical Operations, Dermatology Department, and Professor of Dermatology at the Hospital of the University of Pennsylvania. The study is being supported by a $2.6 million Orphan Products Development Grant from the Food and Drug Administration (FDA). It is evaluating HyBryte treatment for up to 54 weeks in patients with early-stage CTCL. Soligenix previously announced that following 18 weeks of continuous treatment, 75% of patients achieved “treatment success”, which is defined as a ≥50% improvement in the cumulative mCAILS (modified Composite Assessment of Index Lesion Severity) score compared to baseline. In addition, three patients achieved a complete response. We anticipate an additional clinical update in the third quarter of 2025.
Phase 2a Trial in Mild-to-Moderate Psoriasis: Topline Results in 4Q25
Soligenix is currently conducting a Phase 2a trial of SGX302 in patients with mild-to-moderate psoriasis. The company previously announced results from the first two cohorts, which showed a clear biological signal as evidenced by an improvement in PASI (psoriasis area and severity index) score. The study is currently investigating different levels of light intensity/duration to optimize the conditions for psoriasis patients. Once those conditions are optimized, the ultimate goal will be to transition to “at home” use prior to initiating a Phase 3 trial. We anticipate topline results from the last cohort of patients in the fourth quarter of 2025.
Phase 2a Trial in Behcet’s Disease: Topline Results in 3Q25
A Phase 2a proof-of-concept trial of SGX945 (dusquetide) is currently being conducted in patients with Behcet’s Disease (BD). BD is a chronic recurrent multisystemic disease that causes oral aphthous ulcers, genital ulcers, skin lesions, and other pathologies (Mendes et al., 2009). Interestingly, the epidemiology of BD is distributed along the ancient Silk Road from Mediterranean countries (Turkey has 370 cases per 100,000 population), to Middle Eastern and East Asian countries. In contrast, there are very few cases found in Northern Europe (0.64 cases per 100,000 population), North America (0.12-0.33 cases per 100,000 population), Australia, and Africa (Deuter et al., 2007). Thus, BD is an orphan disease in the U.S., however there may be as many as 500,000 people worldwide with the disease.
Dusquetide was previously tested as a treatment for oral mucositis in which it showed biological activity against aphthous ulcers induced by chemotherapy and radiation, thus serving as a proof-of-concept for the treatment of oral ulcers in BD. We anticipate topline results in the third quarter of 2025.
Hypericin Manufacturing Transferred to U.S.
On July 1, 2025, Soligenix announced it has successfully transferred the manufacturing process for synthetic hypericin (the active ingredient in HyBryte and SGX302) from Europe to Sterling Pharma Solutions in the U.S. Soligenix and Sterling have begun cGMP manufacturing of drug product for clinical trials and the long-term plan is to establish a commercial manufacturing agreement.
Conclusion
We look forward to the multiple upcoming catalysts for Soligenix in the second half of 2025. The update on the IIS should provide additional read-through to the FLASH2 trial, which we already have high confidence in being successful based on the robust results announced thus far for the IIS and its similarity to the FLASH2 trial. The psoriasis and BD programs offer additional upside that is not being factored in to the current share price. With no changes to our model our valuation remains at $35 per share.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives quarterly payments totaling a maximum fee of up to $40,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.






