READ THE FULL RANI RESEARCH REPORT
On August 7th, Rani Therapeutics Holdings, Inc. (NASDAQ:RANI) reported second quarter 2025 results. Since our last update, Rani has signed a research agreement with Chugai Pharmaceutical, presented ProGen’s PG102 preclinical data at ENDO and raised additional capital in preparation for a 2H:25 launch of the Phase I RT-114 trial. Rani continues to interact with prospects to sign a deal with an established biopharmaceutical company, which may include upfronts, milestones and royalties.
2Q:25 Operational and Financial Results
Rani reported second quarter results in a press release and Form 10-Q filing with the SEC on August 7th. For the quarter ending June 30th, 2025, no revenues were recognized and operating expense was ($10.5) million and net loss per share for Class A shareholders was ($0.18).
- Research and development expenses totaled $5.5 million, down 10% from $6.1 million mostly due to lower compensation costs, a smaller team partially offset by slightly higher third-party service costs;
- General & administrative expenses were $5.0 million, falling 22% from $6.4 million on lower compensation costs, reduction in workforce and a fall in third party services;
- Net interest expense was ($719,000) vs. ($837,000) due to lower cash balances;
- Non-controlling interest was ($4.5) million vs. ($6.6) million;
- Net loss for Class A shareholders was ($6.7) million vs. ($6.8) million or ($0.18) and ($0.26) per share.
As of June 30th, 2025, cash and marketable securities totaled $10.2 million. This amount compares to the $27.6 million balance held at the end of 2024. Long-term debt was held on the balance sheet at $17.2 million. Cash used in operations and for capital expenditures for the first half of 2025 was ($14.0) million versus ($18.5) million in the same prior year period. During the second quarter, Rani repaid $3.8 million in debt bringing the total to $7.5 million for 2025. Offsetting this outflow was $3.9 million from the exercise of warrants. Following the end of the second quarter, Rani raised an additional $3.0 million from the issuance of 7.5 million shares at $0.40 per share.
Research Agreement with Chugai
A May 19th press release informed investors that Rani had signed a research agreement with Chugai Pharmaceutical Co. on August 13th, 2024. The agreement granted Chugai rights to develop two molecules with undisclosed targets. Since the signing, the partner has demonstrated bioavailability comparable with delivery via the subcutaneous route for both molecules. The partners are now evaluating feasibility of applying Rani’s technology to these antibodies.
Chugai Pharmaceutical is a research and development-oriented pharmaceutical company based in Tokyo, Japan established in 1925. It operates as a subsidiary of Hoffmann-La Roche which owns a 62% stake and trades on multiple exchanges including the OTC under the ticker CHGCY. Chugai has developed and marketed several globally recognized pharmaceuticals, often in partnership with Roche. This includes Hemlibra, Actemra, and Alecensa among other products. Chugai also discovered orforglipron and licensed it to Eli Lilly in 2018.
Endocrine Society’s Annual Meeting (ENDO 2025) Poster
Rani participated in the 2025 Endocrine Society Annual Meeting, which was held in San Francisco July 12th to 15th. As described in a July 14th press release, company scientist Anvesh Dasari, Ph.D. presented a poster entitled Oral Delivery of a Bispecific GLP-1/GLP-2 Receptor Agonist (PG-102) via a Robotic Pill (RT-114) Achieves Bioequivalence to Subcutaneous Injection in Canines. The work presented preclinical data including delivery success rate, oral bioavailability for RP vs. subcutaneous (SC) delivery and weight loss in the canine models. 12.0 milligrams of PG-102, a GLP-1/GLP-2 receptor agonist was delivered into a population of 16 healthy canines. Six were administered PG-102 by SC injection and oral administration of the RP was given to 10 canines. Pharmacokinetic and pharmacodynamic data was measured such as serum drug concentrations, food intake and body weight, safety, tolerability and delivery reliability.
The Rani Pill was successfully delivered in nine of ten subjects and generated oral bioavailability of 111% relative to subcutaneous delivery. Food consumption was inversely correlated to serum drug concentrations for RT-114 and weight loss was comparable between the two arms of the study. Graphic details of the findings are included below.
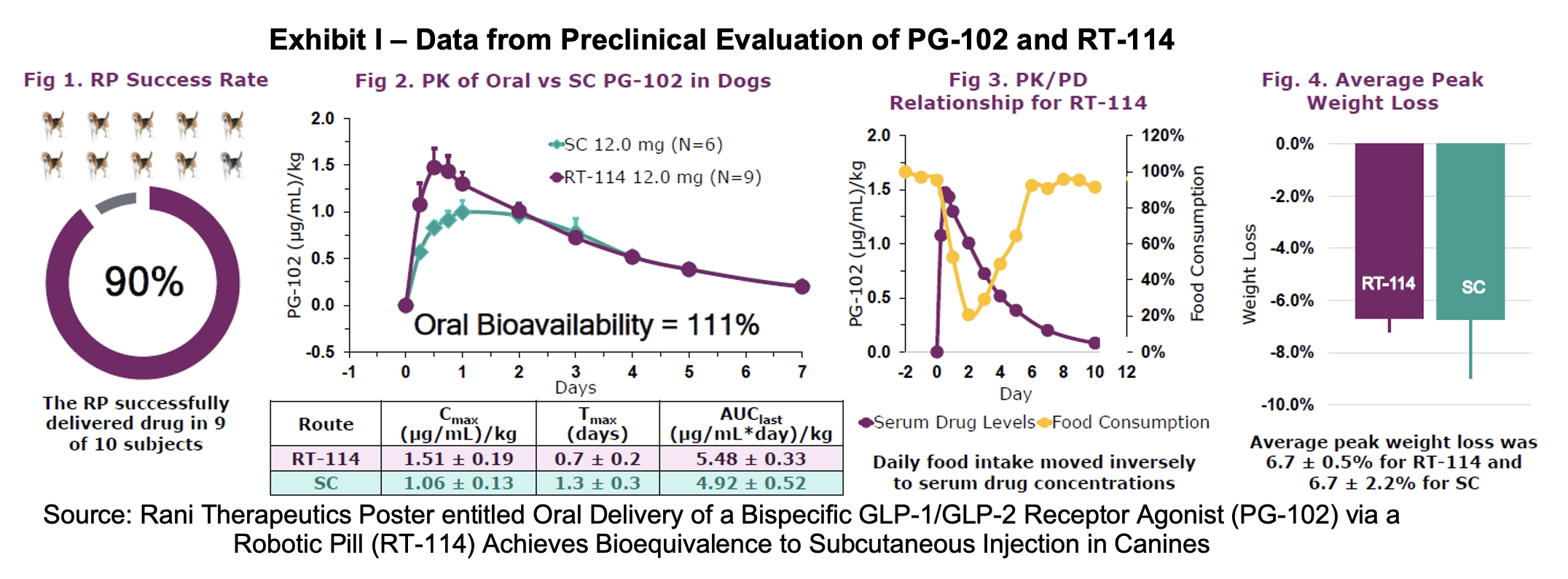
RT-114 AND PG-102 Results
ProGen Collaboration Details
In June 2024, Rani announced a collaboration with ProGen Co., Ltd. to develop an oral obesity treatment combining ProGen’s FC Fusion protein conjugated GLP-1/GLP-2 dual agonist, PG-102, with Rani’s Rani Pill HC. Rani entered into a definitive agreement with ProGen to develop a new product designated RT-114, an oral dose of ProGen’s PG-102. The companies will equally share the costs of development of RT-114 and divide geographies between them. Details of the arrangement between the two companies can be found in the second quarter 2024 Form 10-Q.[1]
Following the deal between Rani and ProGen, the partners have begun a development plan for the GLP-1/GLP-2 dual agonist. As of August 2025, ProGen has completed its Phase I trial for PG-102 with the primary endpoints of safety and tolerability and has started a Phase II study with further details on timing and readout expected later this year. Rani is advancing a head-to-head preclinical comparison of orally administered PG-102 using the RaniPill capsule (RT-114) to 16 healthy canines. Data from this study was presented at the ENDO meeting with details included above.
ProGen Clinical Trials
ProGen’s Phase I trial underway for PG-102 announced preliminary results for the repeat-dose portion (Phase Ic) of its Phase I study. The candidate produced an average weight loss in subjects (N=73) of 4.8% after five weeks of dosing. Subcutaneous PG-102 was well tolerated with subjects able to titrate to the target dose in one month. There were no treatment discontinuations. Additional data was presented at the Asian Association for the Study of Diabetes (AASD) which was held March 28th to 30th. Rani’s Senior Strategic Advisor, Jesper Høiland hypothesizes that the rapid titration may facilitate faster onset, shortening the time between initial administration and target weight loss. It is notable that the 4.8% average weight loss was achieved after five weeks compared to the four to five months required by approved GLP-1 agonists.[2] ProGen is planning a Phase II study of PG-102 that will compare the candidate with placebo in obesity and Type 2 diabetes. Six sites in Korea have been identified and sites outside Korea are also being added.
Rani Preclinical Trials
Rani is running its preclinical study, delivering either a 12 mg dose of PG-102 using RaniPill (N=10) or subcutaneously (N=6). This is estimated to be equivalent to a 60 mg dose in humans. The study is evaluating endpoints including safety, tolerability and reliability of the RaniPill capsule as well as pharmacokinetics, which is serum concentration measured by enzyme-linked immunosorbent assay (ELISA). Body weight and food intake are also being measured.
Milestones
- Preclinical data readout for RT-116 – February 2025
- Research agreement with Chugai Pharmaceutical – May 2025
- Launch of Phase I trial for RT-114 – 2H:25
- End of NASDAQ minimum market value requirement resolution period – October 2025
- End of NASDAQ $1.00 minimum bid price resolution period – December 2025
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] See .pdf page 38 which is the beginning of the Collaboration Agreement listed as Exhibit 10.1
[2] Ryan, D.H., et al. Long-term weight loss effects of semaglutide in obesity without diabetes in the SELECT trial. Nature Medicine. May 2024. See Figure 1.







