In an April 27th press release, Ligand Pharmaceuticals, Inc. (NASDAQ: LGND) announced that it will acquire XOMA Royalty Corporation (NASDAQ: XOMA) for $39 per share in an all-cash transaction. XOMA shareholders will receive a Contingent Value Right (CVR) entitling the holder to receive a 75% of any net proceeds that may result from pending Tremfya litigation with Janssen. The merger requires cash redemption of XOMA’s preferred stock, repayment of loans, along with cash settlements of in-the-money options and certain warrants. The total value of the purchase is estimated to be $739 million. It will be funded through cash on Ligand’s balance sheet and borrowings under an existing credit facility. Management issued a press release, Form 8-K, and slide deck accompanied by a conference call on the morning of April 27th, 2026, to provide the relevant details.
The transaction is expected to close in the third quarter of 2026 and to be immediately accretive to Ligand’s earnings. 2026 revenue estimates increase by $25 million to a range of $270 to $310 million, and earnings per share (EPS) increase by $0.50 per share to a range of $8.50 to $9.50. Incremental operating profit from XOMA in 2026 is expected to be $20 million, offset by a $6 million hit to other income related to reduced capital deployment and acquisition related capital costs. The entire revenue increase will accrue to the Royalties segment. In 2027, earnings per share are expected to be incrementally higher by $1.50.
The acquisition brings more than 100 additional assets into Ligand’s portfolio. This includes seven commercial programs, 14 Phase III or registrational programs, and more than 100 earlier stage assets.
XOMA Revenues
XOMA generated $52.1 million in revenues for 2025, representing an 83% increase over prior year revenues of $28.5 million. Cash receipts were $50.5 million. This was divided into royalties of $33.6 million, with Vabysmo comprising about two-thirds of that amount and milestones and fees of $16.9 million. Analyst estimates[1] call for strong double-digit growth for Vabysmo, Ojemda, Miplyffa, and Xaciato over the next two years.
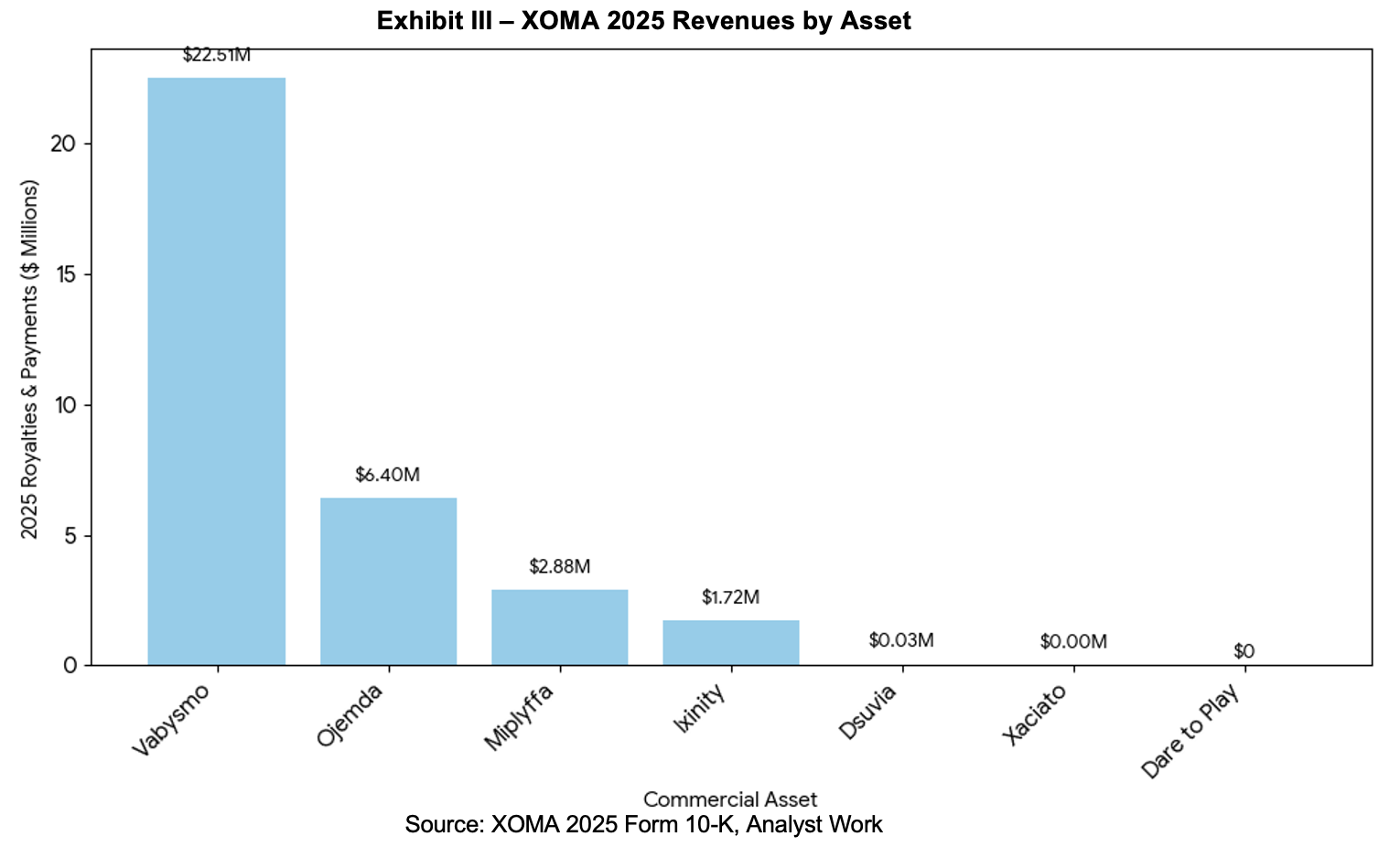
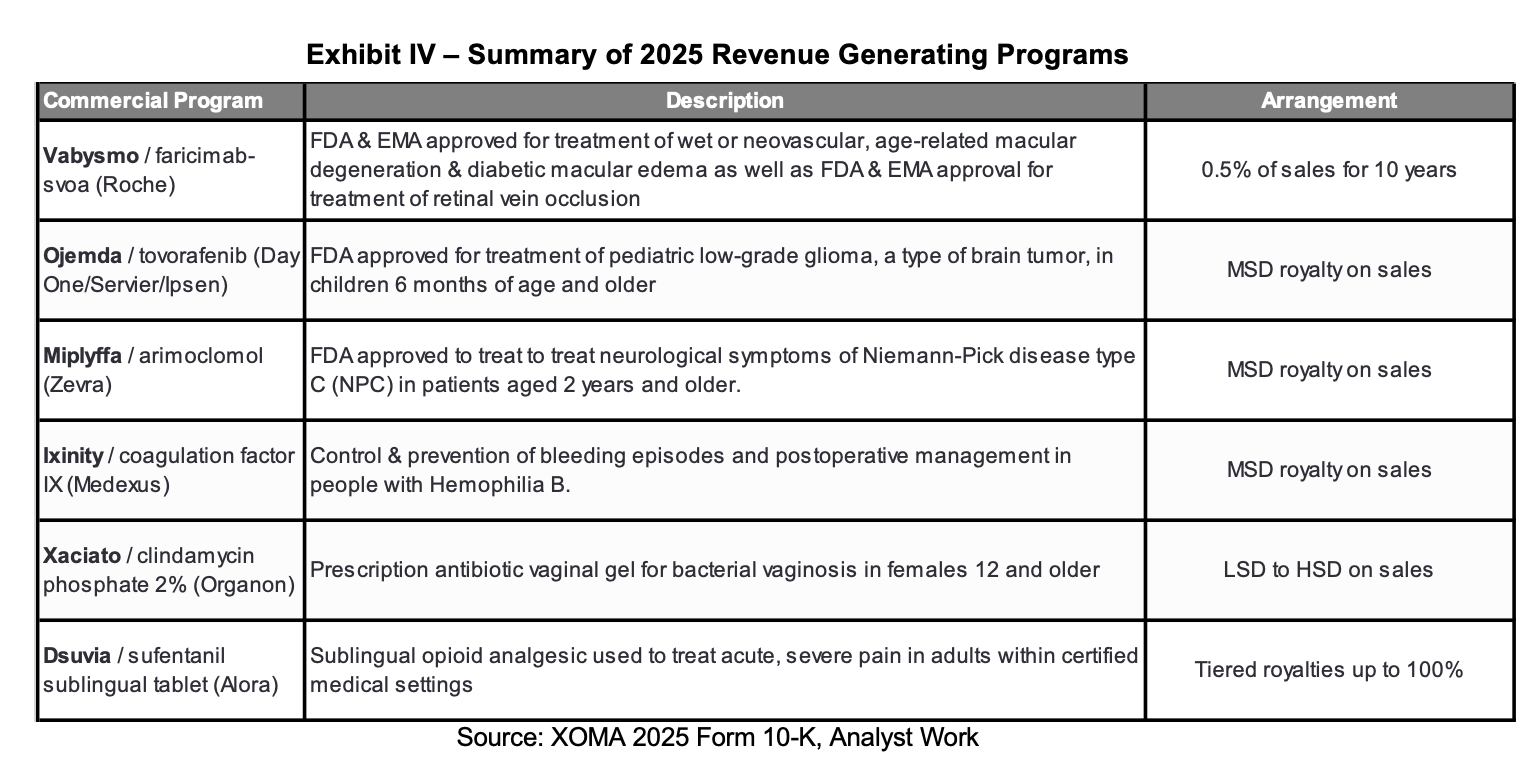
Beyond XOMA’s revenue contribution, Ligand anticipates that it will be able to reduce costs. It has developed an investment team and infrastructure that can accommodate the layering on of additional assets. Management expects that most standalone operating costs associated with XOMA can be eliminated. This is expected to contribute to the anticipated $0.50 in incremental earnings expected in 2026 and the $1.50 of incremental earnings in 2027. Based on the ~21 million shares outstanding at Ligand and applying management guidance, this should equate to about $11 million in 2026 incremental earnings and $32 million in 2027 earnings. Additional details are provided on slide 12 of the presentation.
XOMA also offers tax benefits to the combination. In its 2025 10-K filing, it reported almost $200 million of federal and $23.5 million of state net operating loss (NOL) carryforwards. The use of these tax assets may be severely limited due to anticipated expirations in the 2030s and limitations on use due to change in control. There are also federal research and development (R&D) tax credits of $2.0 million and state (California) R&D credits of about $20 million. While we do not have the data or expertise to estimate the value of these assets, Ligand management believes they are material.
Filspari
Travere’s Filspari was approved in February 2023 to slow kidney function decline in adults with primary immunoglobulin A nephropathy (IgAN) who are at risk for disease progression. Ligand’s partner, Travere, subsequently submitted its supplemental New Drug Application (sNDA) for Focal Segmental Glomerulosclerosis (FSGS) in March 2025. The application was initially given a Target Action Date of January 13th, 2026. However, just prior to this milestone, the FDA extended the review period.
In the weeks prior to the anticipated approval, the FDA made a series of information requests to clarify the benefit of Filspari in FSGS. While the responses were submitted prior to the agency’s decision, there was insufficient time for the agency to properly review the data. As a result, the FDA delayed the Target Action Date by three months, classifying the additional data submission as a Major Amendment. Travere management clarified that the data requested did not include anything related to safety or manufacturing of the drug.
On April 13th, 2026, Travere announced that the FDA had granted full approval for Filspari in adults and children aged 8 years and older with FSGS who do not have nephrotic syndrome. We do not explicitly forecast FSGS revenues in our model, but Ligand does include a component for FSGS revenues in their Pharm Team segment. In its Analyst Day presentation last December, Ligand estimates that the contribution from Filspari for FSGS will be an estimated $4 million in 2026, rising to $40-$45 million in 2030.
Tzield
Sanofi’s Tzield approved in the US to delay the onset of stage 3 type 1 diabetes in young children.
Milestones
- EC approves teplizumab to delay onset of stage 3 type 1 diabetes (T1D) in patients age 8+ – January 2026
- Ohtuvayre NDA acceptance by China NMPA – January 2026
- Data readout for Phase III SELVA trial for QTORIN – February 2026
- Filspari FSGS indication FDA approval – April 13th, 2026
- FDA approved Tzield for delay in onset of stage 3 T1D in stage 2 patients – April 22nd, 2026
- XOMA acquisition announcement – April 2026
- Palvella QTORIN FDA meeting for Phase II study in angiokeratomas – 1H:26
- FDA approval for Tzield in Stage 3 Type 1 diabetes – 1H:26
- Ojemda Phase III full enrollment – 1H:26
- Japan NDA submission for Filspari via Chugai – 2026
- Capvaxive PDUFA date for at risk children & adolescents – June 18th, 2026
- QTORIN NDA submission to FDA for MLM – 2H:26
- QTORIN Phase III trial for cutaneous venous malformations – 2H:26
- QTORIN Phase II trial for clinically significant angiokeratomas – 2H:26
- Ojemda topline data – mid-2027
- Osavampator Phase III topline data readout – 2027
Summary
Ligand is making a bold move with its proposed acquisition of XOMA, which is expected to close in the third quarter of 2026. We expect the $739 million transaction to significantly expand Ligand’s royalty-driven business model, adding a portfolio of more than 100 partnered programs, several of which are already generating revenue, with six assets contributing royalties in 2025. XOMA’s pipeline also introduces a steady stream of potential near- and mid-term catalysts, positioning the combined company for broader and more diversified revenue growth. Management has indicated that the deal is immediately accretive and that the acquired portfolio is growing at a faster rate than Ligand’s legacy assets.
Beyond top-line expansion, the transaction offers meaningful operating leverage. Ligand Pharmaceuticals already has the infrastructure to manage a larger portfolio of royalty-generating assets, suggesting potential cost synergies and improved scalability. In addition, XOMA brings net operating losses and R&D tax credits that could enhance after-tax earnings. Ligand plans to fund the acquisition using a combination of cash on hand and its existing credit facility, while maintaining confidence in its ability to sustain its typical annual investment pace of $150–$250 million.
The XOMA acquisition is not the only recent positive development. In April 2026, the U.S. Food and Drug Administration approved Filspari for focal segmental glomerulosclerosis (FSGS), expanding the commercial opportunity for this important royalty-bearing asset. It also approved Sanofi’s Tzield in the US to delay the onset of stage 3 type 1 diabetes in young children, expanding the utility of the drug. Together, these developments reinforce Ligand’s strategy of building a diversified, high-margin royalty portfolio. We expect to find additional clarity on integration plans, capital allocation, and updated growth expectations when the company reports its first-quarter 2026 results on May 7th, 2026.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] Sourced from Evaluate, Ltd.









