READ THE FULL HURA RESEARCH REPORT
Operational and Financial Results
On August 15th, 2025, TuHURA Biosciences, Inc. (NASDAQ:HURA) reported second quarter 2025 financial and operational results and filed its Form 10-Q with the SEC. The company has begun its IFx-2.0 Phase III trial in Merkel cell carcinoma (MCC) and has completed the acquisition of Kineta along with its newly named lead asset TBS-2025. TuHURA recently launched a Phase Ib/II study for IFx-2.0 in MCC patients not eligible for the Phase III trial which is expected to read out in 1Q:26. Other important milestones included presentation of clinical data at the American Association of Cancer Research (AACR) annual meeting in Chicago and the raise of additional capital. Below, we summarize TuHURA’s quarterly financial results.
TuHURA generated no revenues in 2Q:25 and expended $9.9 million on operational activities related to advancing IFx-2.0 and other programs producing a net loss of ($9.5) million or ($0.21) per share. For the quarter ending June 30th, 2025 and versus the same prior year period:
- Research & development expense totaled $4.9 million increasing due to greater expenses for the IFx-2.0 program, preclinical research related to IFx-3.0 and myeloid derived suppressor cell (MDSC) initiatives and higher salary, personnel and facilities costs.
- General & administrative expense totaled $4.9 million, due to increases in non-cash stock compensation, merger transaction costs and expenses related to being a public company;
- Other items included $322,000 in grant income related to Kintara’s REM-001 asset and reimbursements from Health and Human Services;
- Interest income was $29,000 related to income paid on cash balances held;
- Net loss was ($9.5) million or ($0.21) per share.
As of June 30th, 2025, TuHURA held $8.5 million in cash on its balance sheet. 1H:25 cash burn was ($11.0) million while cash generated from financing was $8.2 million which consisted of warrant proceeds and issuance of common stock partially offset by stock issuance costs and transaction and liability payments related to Kintara. Following the end of the quarter and in August 2025 we expect TuHURA to receive an additional $4.7 million in funding.
Private Placement
In early June, TuHURA entered into a $12.6 million equity financing transaction in a private offering with Paulson Investment Company. The arrangement was structured in tranches attached to development milestones related to the advancement of the IFx-2.0 program and the close of the Kineta merger. Four equity investments were made in June totaling approximately $9 million in shares. Funds were disbursed in equal amounts at: the initial closing, the lifting of the partial clinical hold on IFx-2.0, the start of the Phase III IFx-2.0 trial and satisfaction of all material conditions for closing the Kineta merger. A total of 4,759,390 shares were offered at $2.65 per share. Warrants were attached with an exercise price of $3.3125 and a five-year life. There is an additional $3.7 million related to the offering funded by December 31st, 2025. This share purchase also includes warrants with a $3.3125 exercise price and a five-year life. Management expects this amount to close in August 2025.
Close of Kineta Merger
TuHURA closed its merger with Kineta on June 30th, 2025 and renamed its anti-VISTA asset TBS-2025. In addition to TBS-2025, the deal brought worldwide patents, patent rights, patent applications, product and development program assets, technical and business information and other rights. TuHURA anticipates that it will realize many synergies from owning both its IFx-2.0 and anti-VISTA platforms. The company executed several consulting agreements with former Kineta executives to help guide the transition to TBS-2025 and start of the related Phase III trial.
Phase III IFx-2.0 Trial in MCC
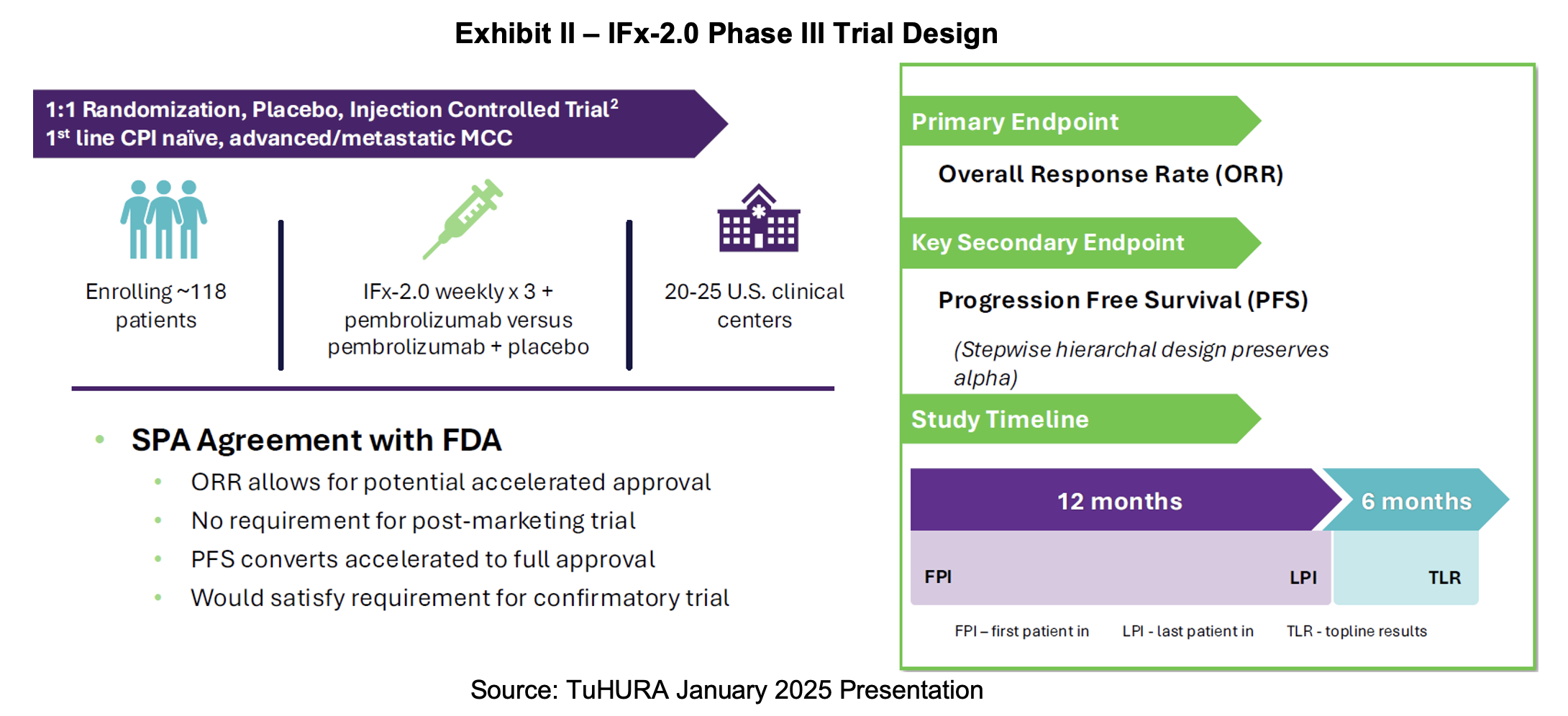
TuHURA launched its pivotal Phase III study for its IFx-2.0 candidate in Merkel cell carcinoma (MCC) in June following the removal of the partial clinical hold related to chemistry, manufacturing and controls (CMC) requirements. Achieving this milestone freed up the associated $2.23 million of funding from early June’s private placement. In the second quarter report, TuHURA set forth two related milestones for the study. Management will provide an update on enrollment progress by year end and distribute topline results by 2H:26.
IFx-2.0 will pursue a new drug application using the FDA’s accelerated approval program under a special protocol assessment (SPA). The trial was designed with the input of the FDA’s Oncology Center of Excellence (OCE). Accelerated approval allows the sponsor to use surrogate endpoints that predict clinical benefit and is appropriate when pursuing serious indications that lack other therapies. In most cases, an accelerated approval will require post-market confirmatory trials to verify the clinical benefit. However, in this case, the FDA has indicated that secondary endpoints that demonstrate clinical benefit may be used. If successfully achieved, the trial may satisfy the requirements for full approval.
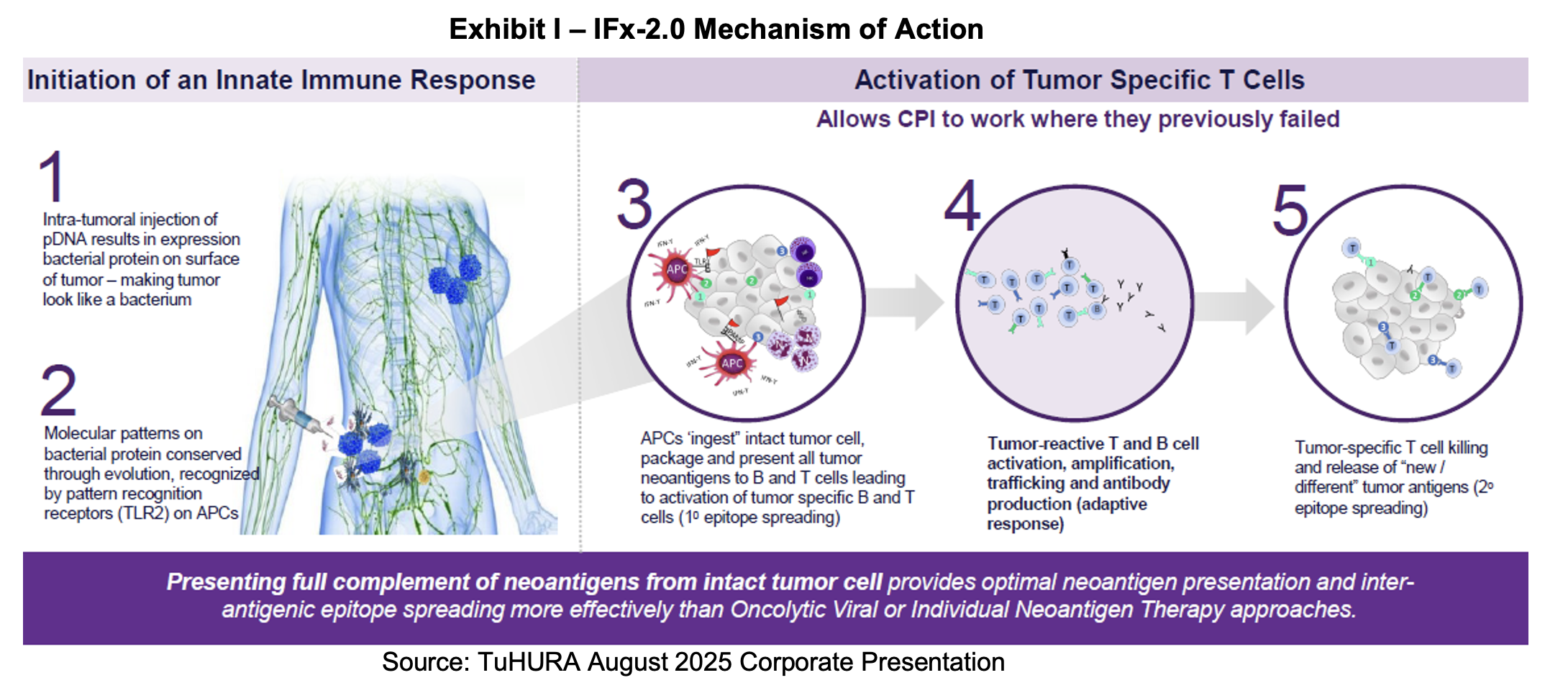
The trial is designed as a 1:1 randomized, placebo and injection-controlled trial in first-line, checkpoint inhibitor naïve, advanced metastatic MCC. It plans to enroll 118 subjects at 20 to 25 clinical centers in the United States. IFx-2.0 will be administered three times per week in combination with pembrolizumab vs. placebo with pembrolizumab. The primary endpoint will be overall response rate (ORR) which will allow for accelerated approval. The secondary endpoint, which indicates clinical benefit is progression free survival (PFS). If this endpoint is achieved, it will allow for full approval and satisfy the requirement for a confirmatory trial.
For the primary endpoints to support an accelerated approval, ORR needs to be 75% compared to the 50% which is the historical rate from other trials. For full approval, based on a continuation of this same trial, PFS has to achieve a 50% or better result. The trial is expected to start in the second quarter of 2025 with enrollment lasting 12 months. ORR will be measured at six months, which suggests that top line results can be available 18 months after the trial begins. Accelerated approval will allow a rolling review and potential approval within six months of topline data.
TBS-2025 Phase I/II Clinical Trial in Solid Tumors
The TBS-2025 (formerly known as KVA12) program is collaborating with Merck through the latter’s contribution of pembrolizumab (Keytruda). It seeks to determine the safety and efficacy of TBS-2025 alone and in combination with the PD-1 inhibitor in a Phase I/II clinical trial. A trial summary is posted on clinicaltrials.gov under NCT05708950. The primary endpoints are adverse events. Secondary endpoints include pharmacokinetics, immunogenicity, tumor response as per Immunotherapy Response Evaluation Criteria in Solid Tumors (iRECIST) standards and ORR. The study will be conducted in four parts: The first two will focus on dose escalation (single-agent and in combination), and the second two will concentrate on dose expansion (single-agent and in combination). Up to 10 dose escalation cohorts will be evaluated in Phase I and maximum tolerated dose will be identified. Exploratory endpoints will examine receptor occupancy, chemokine and cytokine levels in blood, immune cell populations in blood and VISTA expression in the tumor pre-and post-treatment. Up to 60 adult patients with advanced or metastatic solid tumors that have progressed or have been non-responsive to standard-of-care therapies are eligible to enroll.
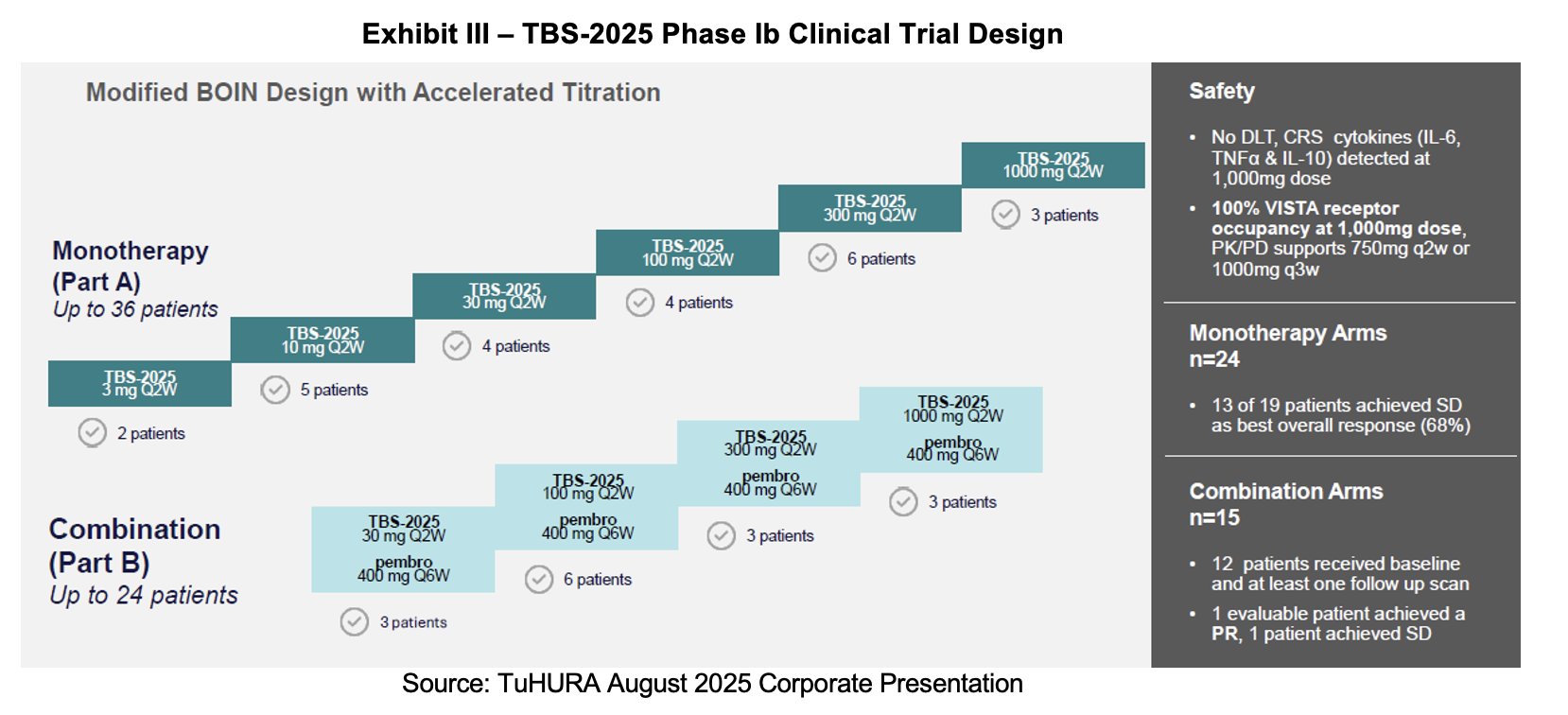
TuHURA is now in the process of closing out the Phase I study report for TBS-2025 and is targeting the Phase II trial start towards the end of 2025. Before it can begin, it must deliver the clinical trial protocol to the FDA, hold its end-of-study meeting with the FDA where it will validate the recommended dose. TuHURA will also select the menin inhibitor that will be used in combination with TBS-2025.
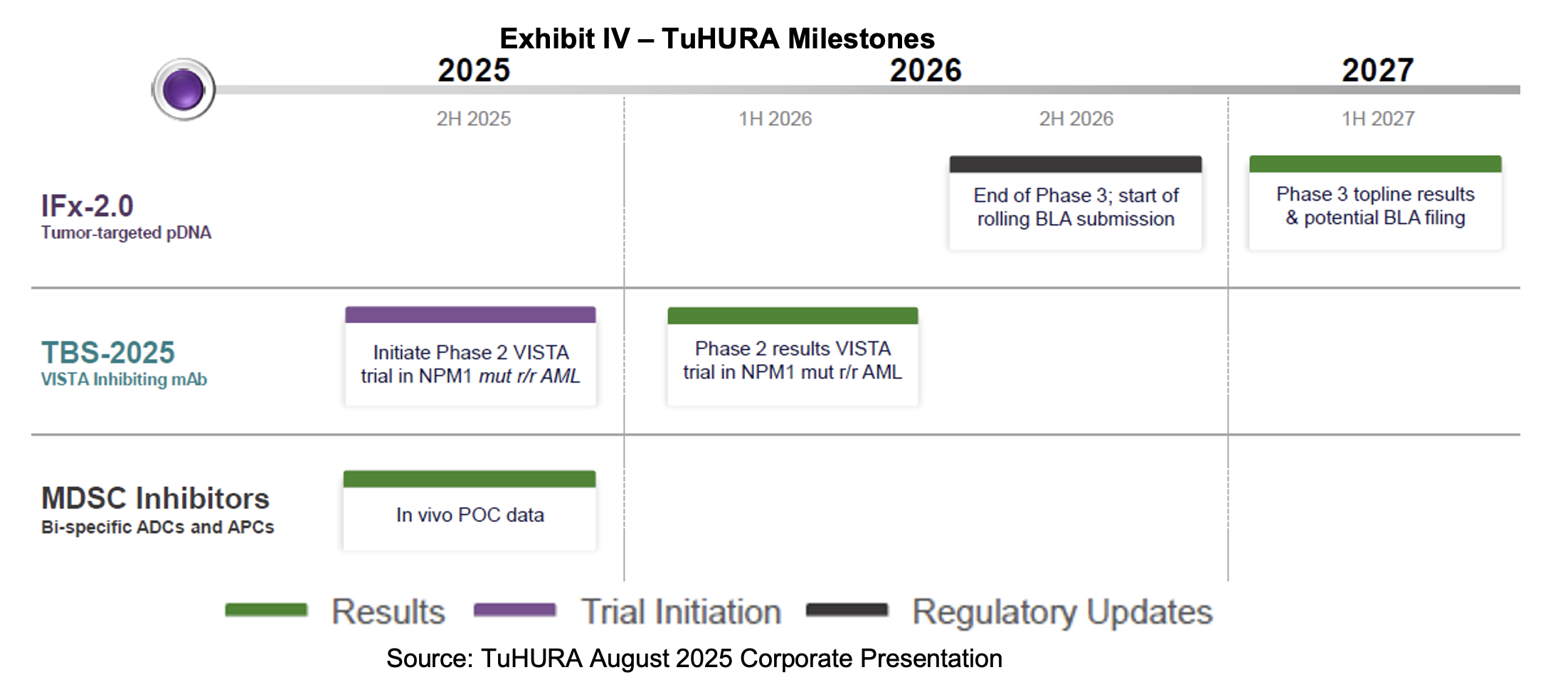
Upcoming Milestones
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.






