READ THE FULL CING RESEARCH REPORT
Cingulate, Inc. (NASDAQ:CING) reported second quarter 2025 results on August 19th, 2025. No revenues were recorded and operating expense of $4.7 million was recognized. The company has now submitted its new drug application for CTx-1301 and is looking ahead to October for an anticipated acceptance by the FDA. Cingulate also saw changes in the management ranks as the company’s focus shifts to pre-commercialization activities and communication at investor and medical conferences. A purchase agreement was signed with Lincoln Park Capital, which is expected to augment the in place at-the-market (ATM) facility and support the crescendo of activities into anticipated approval in the middle of 2026.
2Q:25 Financial and Operational Results
Cingulate reported second quarter results in a press release and Form 10-Q filing with the SEC on August 19th. For the quarter ending June 30th, 2025, the company reported a net loss of ($4.8) million or ($1.09) per share. For 2Q:25 versus the same prior year period:
- Research and development expenses were $2.7 million, up 44% from $1.9 million, attributable to an increase in clinical operations and regulatory costs partially offset by a decline in manufacturing costs. The increase in clinical operations costs was attributable to the close-out and recognition of analytical activities required for NDA submission of CTx-1301. Regulatory costs were up as the team prepared for the pre-NDA meeting with the FDA and the NDA submission;
- General & administrative expenses rose 47% to $1.9 million from $1.3 million on account of higher legal and professional fees;
- Net interest and other expense were ($139,000) compared to ($3,000) related to interest incurred on outstanding notes payable offset by interest earned on cash balances;
- Net loss was ($4.8) million vs. ($3.2) million or ($1.09) per share in 1Q:25.
As of June 30th, 2025, cash totaled $8.9 million. This amount compares to the $12.2 million cash balance held at the end of 2024. Cash burn was ($9.4) million for the first half, partially offset by $6.1 million largely consisting of financing cash contributions from the ATM Agreement with H.C. Wainwright and the Lincoln Park Purchase Agreement. After quarter end, Cingulate raised an additional $690,000 through the ATM and Purchase Agreement.
CTx-1301 FDA Submission Timeline
Safety results from Cingulate’s Phase III studies were submitted to the FDA this spring. A March 4th press release provided a preview of the safety data from two Phase III pediatric and adolescent studies. This included a fixed dose study, a dose optimization study, a food-effect study with healthy adults, using a single 50 mg dose of CTx-1301.
A May 14th press release announced that the notes from the meeting had been received and that the NDA was on track for submission this summer. These minutes are important as they document the key discussion points, any agreements reached and the FDA’s recommendations. The notes serve as an official record of the meeting and provide a reference throughout the drug development and approval process.
A summary of the FDA’s minutes as provided by Cingulate is below.
- FDA agreed to a post-NDA approval commitment of additional stability data for CTx-1301;
- Available nonclinical safety data appears to support a filing;
- CTx-1301 may rely on Focalin XR’s efficacy and safety data;
- Cingulate’s proposed approach to the integrated safety summary and integrated summary of efficacy appears reasonable.
On August 6th, Cingulate announced that it had submitted its NDA to the FDA for CTx-1301. It expects to hear whether or not the submission has been accepted within 60 days of submission. If accepted, this sets up the application to potentially receive approval by mid-2026.
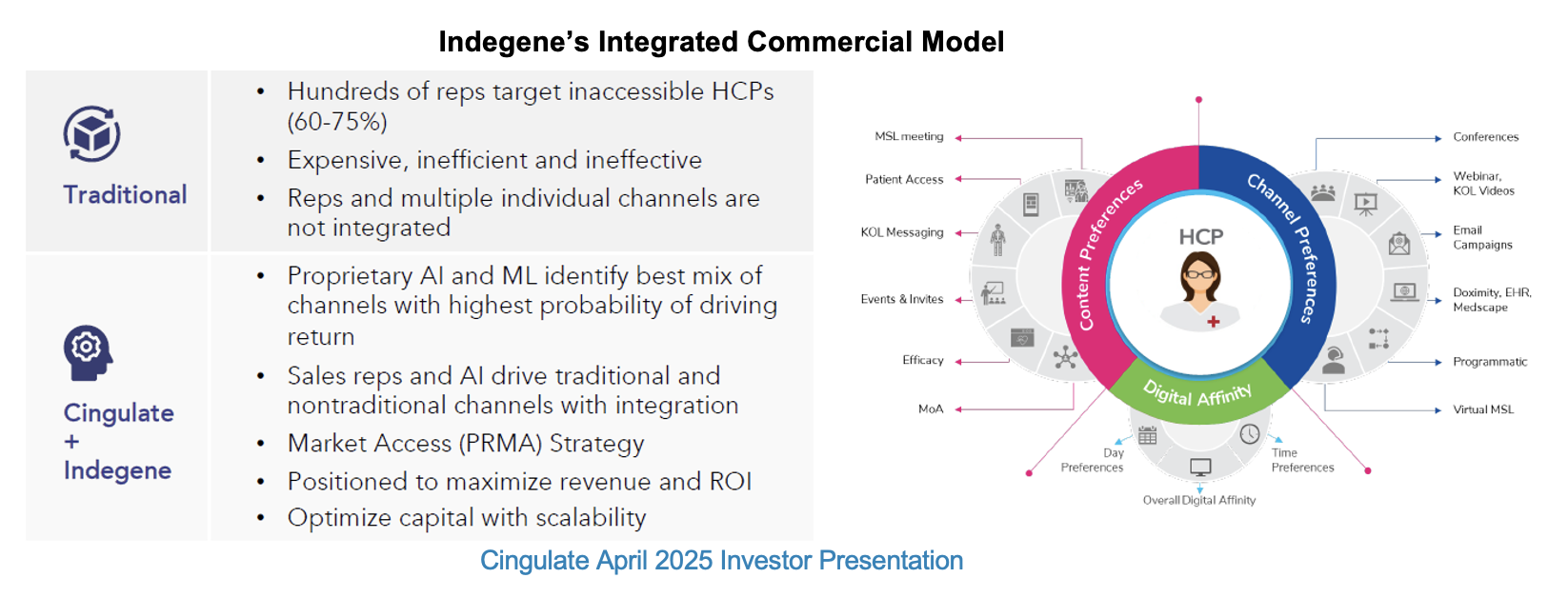
Assuming normal review and approval timelines, commercialization could start as soon as next year. Cingulate will work with Indegene[1] to commercialize the product in the United States and may also pursue a co-promote that would leverage the strengths of multiple parties. Outside the US, Cingulate is looking for partners to commercialize CTx-1301. Beyond international sales, the goal of these relationships is to obtain upfront amounts that will support CTx-1301’s launch in the United States. While the team has not affirmed any specific discussions, we believe that they have had at least initial talks with prospective global partners.
Management Changes
On August 15th, Cingulate provided a management update informing investors of the appointment of CFO Jennifer Callahan as interim CEO and board member Jay Roberts as Executive Chairman. Shane Schaffer is on administrative leave in connection with ongoing legal matters unrelated to the company and its operations. While any change in management calls for a review of the team, the heavy lifting for the next year has been accomplished with the successful submission of the NDA for CTx-1301. We expect the team to begin a measured ramp of pre-commercialization activities.
Objectives Over the Next Twelve Months
With the submission of CTx-1301 now complete, we expect to see acceptance of the NDA in early October. In parallel with the regulatory process, Cingulate management and related parties will participate in investor and medical conferences to communicate the value of its product. CEO Jennifer Callahan, Vice President of Corporate Communications Thomas Dalton and co-founders Dr. Matthew Brams and Dr. Raul Silva are all participating in the messaging effort on behalf of the company. A key opinion leader in the attention-deficit/hyperactivity disorder (ADHD) space and advisor for Cingulate, Dr. Ann Childress, will also present at medical conferences over the next several months. This includes the American Academy of Child and Adolescent Psychiatry (AACAP) conference in October where she will be presenting on behalf of the company.
Pre-commercialization activities have also moved to the forefront. Cingulate has announced its non-binding arrangement with Indegene to commercialize CTx-1301 in the United States on a fee for service basis. In preparation, management is now conducting market access research to better understand how its time release version of dexmethylphenidate can be successfully adopted, reimbursed, and used by patients. The company will also soon sign its commercial supply agreement and begin the manufacturing of process validation batches. The resulting drug product will be saleable and can be used to fulfill pre-launch inventory.
SUBSCRIBE TO ZACKS SMALL CAP RESEARCH to receive our articles and reports emailed directly to you each morning. Please visit our website for additional information on Zacks SCR.
DISCLOSURE: Zacks SCR has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing research coverage for a period of no less than one year. Research articles, as seen here, are part of the service Zacks SCR provides and Zacks SCR receives payments totaling a maximum fee of up to $50,000 annually for these services provided to or regarding the issuer. Full Disclaimer HERE.
________________________
[1] See our initiation for further details on the Indegene Joint Commercialization Agreement.